One of the goals of MetabERN is to facilitate access to better healthcare and treatment to patients with Inherited Metabolic Disorders, regardless of their country of origin. This ambitious goal will be attained by fulfilling 9 SMART objectives, corresponding to the different Work Packages (WPs) detailed in the figure below.
WORK PACKAGES
MetabERN specific, Measurable, Attainable, Realistic, Timely (SMART) OBEJECTVES
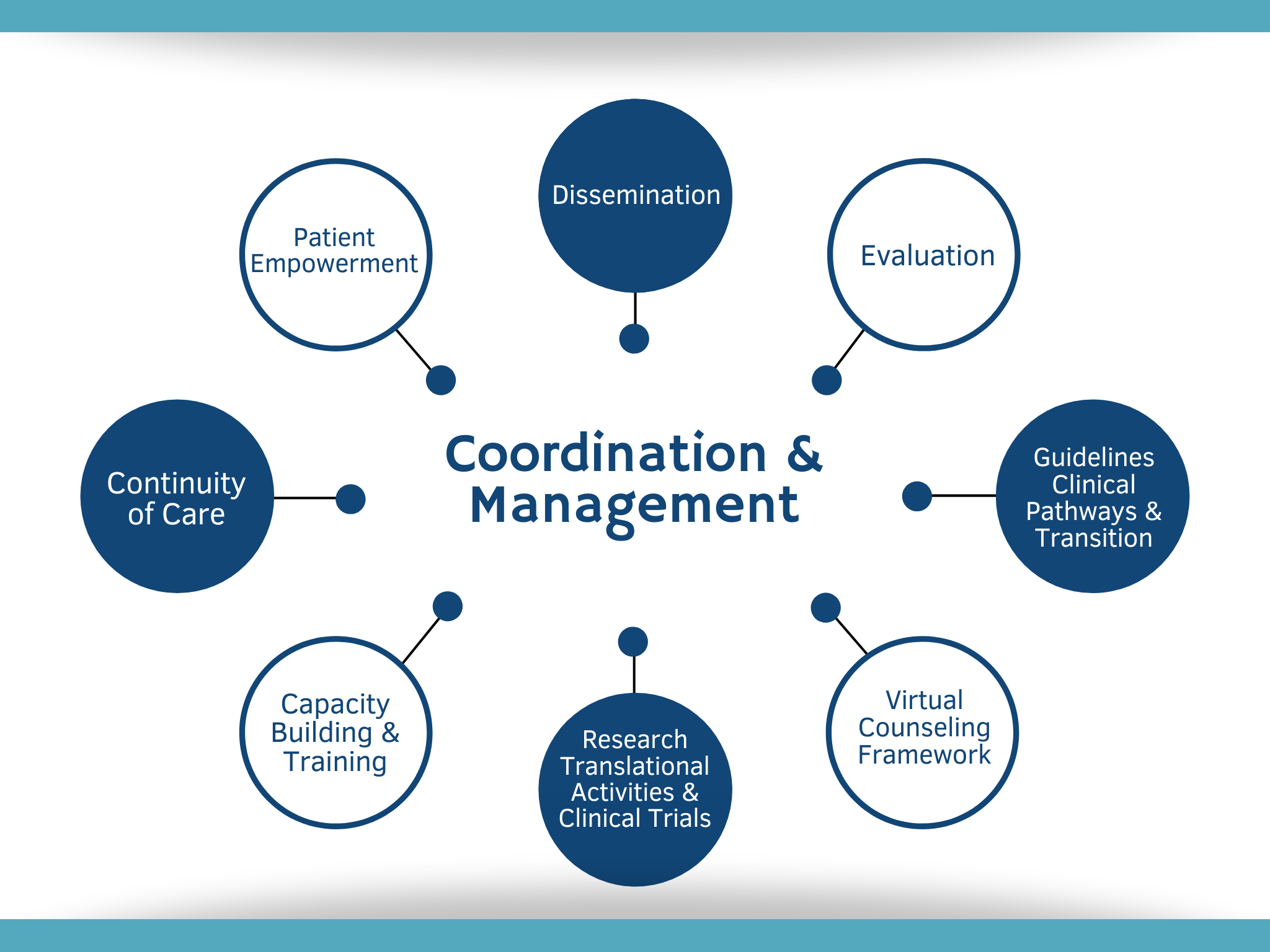
For each WP, methods and means to ensure that the objectives are met within five years have been defined. Click on the list below for further details.
OBJECTIVE: This Work Package is developed for the good management of the project, including the submission of interim and final reports, and for administrative and accounting activities, as well as the disbursement of funds for approved activities. The scope of the work includes activities for the day-to-day monitoring of the project progress.
METHODS:
Define a detailed structure to facilitate the organization of the network.
Instruct the full members on the timeline, milestones and deliverables.
Distribute duties according to a survey already performed during the preparation of the application.
Create a draft calendar of the MetabERN meetings and activities to be shared with all stakeholders.
Prosecute of the collaboration with RD Action to optimize the management of patients across ERN expertise.
Maintain constant contact with the EC and the other coordinators to harmonize and integrate expertise and impact of the EU rare diseases policy.
Perform a survey on the potential funding to identify possible sources of continuity.
Finalize the MetabERN statute in order to ensure compliance with and adherence to the rules and responsibilities of HCPs participating in MetabERN
DELIVERABLES
MetabERN Board meeting
Adopted/ signed MetabERN statutes
Meeting with coordinators of overlapping ERNs and strategy on overlapping ERNs
Meeting of CORE-ERN working group
Annual report
Coordinator
OBJECTIVE: MetabERN activities will be supported through presence at different levels, including interaction with EU institutions, scientific conferences, as well as on- and offline publications. Patient education and empowerment will be achieved through wide knowledge dissemination by patient support group education, newsletters, videos, patient conferences, teleconferences, educational presentations on the web. The “physical” and “virtual” presence of MetabERN at these sites will also increase public awareness, which can have direct spinoffs, like early patient detection, social or psychologic support, but also bring extra opportunity for new scientific ideas, public interest, out of the box thinking in research on different MetabERN topics and could offer more opportunity for additional funding. SSIEM and the Journal of Inherited Metabolic Disease (JIMD) as Official Journal will be strongly involved.
METHODS
Organisation of meeting and teleconferences to find the most efficient and creative ways to communicate between centres and their supporting networks
Traditional media outreach including a press release for the initial launch of the project and publication of editorials in relevant peer reviewed outlets such as the Journal Inborn Errors of Metabolism
Production of dissemination materials such as the production of MetabERN-branded leaflets for hospital and academic centers
Profiling of MetabERN in relevant medical congresses: sub-networks submit an information poster on their goals and organization at the next Society for the Study of Inborn Errors of Metabolism meeting, which is also the world conference for IEM next year. The editorial team of the Journal Inborn Errors of Metabolism will write an editorial on MetabERN.
Development of online dissemination content for EU IT Platform that will include extensive information on IMDs.
Set up of social media accounts (Facebook, Twitter)
Dissemination of general information addressed to a lay audience and ad hoc information for patients and their families as well as for healthcare professionals
DELIVERABLES
Develop dissemination channels (website, social media, newsletter)
Patient engagement: attendance of 1 patient meeting per Member State
External dissemination: press release, article in medical journal, MetabERN brochure for patients
Coordinator
Patient Representative
OBJECTIVE: This work package has three major aims: (1) to assist the Coordinator, Management Board and Advisory Board with monitoring progress, (2) to ensure that interactions between stakeholders involved in the care for patients with IMDs are transparent and credible, and (3) to evaluate the impact of MetabERN activities on patient care.
METHODS:
Development of a robust internal evaluation framework for MetabERN to monitor implementation and progresses across Work Packages and within each Sub-Group.
Development a robust and reliable set of simple outcome measures to evaluate the matrix of tasks across different WPs and disease-oriented sub-networks.
Establishment of the MetabERN Conflict of Interest Policy
Measurement of the impact of the educational activities through surveys on satisfaction which will examine whether these needs have been met by the MetabERN activities
DELIVERABLES
Adopted and signed conflict of interest policy
Identification and guidelines of outcome measures and evaluation tools
Survey on patients’ needs
Survey on HCPs’ needs
Coordinator
Patient Representative
OBJECTIVE: By providing guidelines and clinical pathways, MetabERN can improve the diagnostic and treatment abilities of medical centres. Wide dissemination of these through easy-to-access, electronic platforms and pocket-book guides will allow for standardisation of care and growing awareness of IMDs. Guidelines will be based on best available scientific advice and written by MetabERN’s disease sub-groups so they are tailored to specific IMDs.
METHODS:
WP4 will work in close cooperation with WP1, WP3, WP6, WP7 and WP8.
Evaluation of already existing GLs/CPs to assess success factors that could be replicated in other disease areas.
Definition of the specifications:
for an interactive electronic tool for the development of GLs/CPs.
for evaluation of the impact of GLs/CPs on disease management and improvement of outcomes.
Electronic tools will be available from the EC for this process and their usefulness will be investigated. (Consultations with all WP leaders mandatory)
DELIVERABLES
Report on available guidelines for clinical pathways development
Guidelines for clinical pathways development
Coordinators
OBJECTIVE: The aim of this WP will be to establish a common electronic platform for both counseling of individual patients, physicians and/or scientists and group discussions with varying partners. Different options for virtual data-sharing and guidelines on questions of intellectual property will be developed as to provide a coherent framework for further projects on second opinion and the ultimate creation of cross-border multidisciplinary teams.
METHODS:
Aim to establish a common electronic platform in cooperation with other WPs, in particular WP1, WP2, WP4, WP7 and WP9 to overcome limitations of time and distance
Virtual counseling is based on the use of the CPMS.
Survey of the current used hard- and software by participating partners to develop a reference baseline.
Establish formal policy on intellectual property and handling of unpublished data.
DELIVERABLES
Report on data exchange strategy
Policy on technical solutions and regulatory measures for data exchange
Strategy on intellectual property
Patient Representative
OBJECTIVE: Another key component of MetabERN will be research. Several issues currently obstruct patient diagnosis and treatment including delayed diagnosis, limited availability of treatments and clinical trials for new treatments that are poorly designed and carried out. We recognise the limiting factor will be funding, so a grant office will be established to support the research activity.
METHODS:
The core elements that will be addressed by WP are
The delay of diagnosis,
The limited availability of treatments for a number of diseases,
The design of clinical trials failing to generate reliable data
The paucity of reliable biomarkers for the evaluation of drug safety and efficacy.
Problem of funding
In collaboration with patient organisations, we identified the needs in research by performing a survey to present a picture of the research capacities and activities at the European level.
The results of the survey were published in May 2019 in the scientific paper Research activity and capability in the European reference network MetabERN, including recommendations on the priority setting for the research agenda in the area of IMDs.
Visit our publications page to find the study.
Establishment of a grant office
DELIVERABLES
Survey on research capacity/ interests of MetabERN members
Report on advancements in selected fields of IMD research (e.g. diagnosis, screening)
Creation of grant office
Coordinator
OBJECTIVE:
This MetabERN work package will focus on the identification of training and teaching needs as well as already available capacities within the network and how they can be used to develop a comprehensive programme for the benefit of participating HCPs, young PhD students as well as patients and their families. One particular focus will be put on capacity-building in affiliated members as well as those centres currently underdeveloped in certain areas.
METHODS:
During the first year, WP7 will focus on the identification of teaching and training programmes already available to HCPs and patient organisations within the network.
The WP will develop a report on those options available, including target groups, types of programmes, etc. and provide a short report on their findings to the MetabERN Board, Medical Executive Board, Patient Board and the Advisory Board for consideration.
Based on the findings and interests of MetabERN members, a strategy on the provision of training programmes for MetabERN members will be developed in close collaboration with the SSIEM Academy.
An overview of available programmes will be disseminated through the website as well as through regular updates on upcoming programmes throughout the year.
The WP will work closely with WP2, WP5 and WP7
DELIVERABLES
Report on available training and teaching programmes
Strategy of training programmes in collaboration with SSIEM Academy
Coordinator
Patient Representative
OBJECTIVE:
MetabERN aims to harmonise diagnosis, screening, prevention and treatment of IMDs across Europe, and improve longitudinal care of patients with IMDs, by building multinational cohorts of patients with rare disorders, so that experience can be shared and guidelines developed for a more unified and structured follow up. Such multinational cohort data will pave the way for clinical trials of novel therapies and other research interests.
METHODS:
To ensure continuity of care across Member States, during the first year, we will draft a strategy to ensure that also for the MS not still part of MetabERN a contact is established with affiliated members identified according to national rules.
This will be important to ensure:
inclusiveness and the contact with those potential HCPs that could be admitted upon their Member States’ endorsement.
access to expertise also to those patients living in non MetabERN MS, showing how important is to fulfil the spirit of service of MetabERN.
The contacts will be established via multiple channels, including patient associations, that will help identify those centres that could benefit from the Network.
Make an inventory of diagnostic tools that are available in Europe
DELIVERABLES
Report on certified diagnostic technologies and services in Europe
Coordinators
OBJECTIVE:
Improving patient-centred health-care, patients’ involvement and integration of patients in our Network
METHODS:
By working together with the HCPs in the Subnetworks, patients should have a voice in every aspect of the working of the Network. The Steering Committee is leading the way by setting out the path for patients within MetabERN.
DELIVERABLES
- Making recommendations for patient-centred healthcare including in clinical trials and broader industrial strategies
- Implementation of patient involvement and integration in the Network.
Steering Committee
 Copyright ©2017-2021 all rights reserved
Copyright ©2017-2021 all rights reserved 